I had a brief discussion with someone at the International Behavioral Ecology meetings about evidence in bats for prepared learning–the phenomenon that animals learn some associations faster than others. More importantly, the notion here is that animals learn things faster when those lessons would be most common and necessary in the environments in which they evolved.
The classic example is that a rat quickly associates feeling sick with a novel flavor of food, but it’s slower to associate feeling sick with a novel sound or sight [1]. Conversely, the rat learns faster to associate an electric shock with a novel sound or sight than with eating a novel flavor.
Most psychologists consider this quite special and call it an “adaptive specialization for learning” and even find it quite controversial or unbelievable [2]. I, on the other end of the spectrum, think most learning probably works this way. We just haven’t done enough studies on this topic which make that clear, because it’s difficult to show.
What’s truly impressive is that one form of prepared learning has been experimentally evolved in fruit flies [3].
Two years ago, Dunlap and Stephens did a great experiment [3]. They created artificial worlds where flies were selected to learn to avoid an egg-laying site that was previously paired with quinine (which had been done before); and in addition to this, they created selection that favored learning some associations more readily than others.
To do this, they created two egg-laying sites that differed in both smell and color. In some worlds, the association of quinine with color was the best predictor of which type to avoid; in others the association of quinine with odor was most adaptive. That is, their experiment made one type of cue (odor or color) a more reliable indicator of flavor of quinine, which indicated that your eggs would not survive. In world 1, quinine was always indicated by the color of the food dish and odor was only half as reliable. In world 2, quinine was always indicated by an odor and color was only half as reliable. In worlds 3 and 4, both were either reliable or unreliable. For 40 generations of evolutions, they allowed 10 populations of flies to evolve in each world. And yes, they found that when only odor associations were 100% reliable, the flies evolved increased sensitivity to learning the quinine–odor experience and reduced sensitivity to learning quinine–color. And the same was true for the other three worlds. In other words, these flies were born prepared to learn some things faster than others.
So there’s another reason to throw “nature vs nurture” out the window. In my opinion, natural selection doesn’t just give you the ability to learn, it gives you adaptively biased learning. Nature determines how you respond to nurture.
I really don’t think it could be any other way. You can’t build a general associative learning machine that pairs any information with any outcome based on repeated pairings. You can only build a machine that takes certain selection of sensory inputs and pairs those with other selected inputs to produce outputs. If A + B, then do C. But an animal has countless number of sensory inputs during every second of it’s life. Learning is always based on rules (about how to learn and what to learn) that are not themselves learned. So in some sense, it can be argued that all learning is in some degree prepared; it’s just a question of how much.
We can also see evidence for prepared learning in how animals learn today. Evidence for this comes from the niche-specific cognitive strategies hypothesis, put forth by York Winter, which predicts that animals feeding on stationary food like flowers (or that cache food in different locations [4-5]) will be faster to learn rewarded locations, whereas animals that feed on very mobile food (like insects) will be faster to learn rewarded sensory cues (like a visual cue for a predatory bird or an echo-acoustic shape cue for a predatory bat).

The neotropical bat, Glossophaga soricina (shown left) feeds on both insects, fruits, and floral nectar, but it possesses morphological and cognitive adaptations for flower-feeding. These bats will revisit the same flower as many as 30 times in a single night [7]. As expected from the niche-specific cognition hypothesis, Glossophaga has an excellent spatial memory that can overshadow sensory cues [8-9]. The bats can learn a rewarded location instantly, but they cannot ignore location and learn only using shapes. Even when spatial cues to the location of food become unreliable, Glossophaga has great difficulty in learning to ignore location and use shape cues instead [8]. In a study by Stich and Winter [9], bats were presented with choice between two very different shapes, one rewarded and one unrewarded, and the arrangement of shapes were swapped from left to right (see image below).
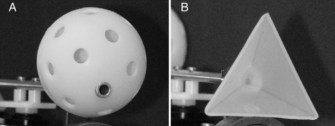
So all the bats had to do was learn to associate a shape with food. According to associative learning theory, this should be pretty trivial.
But here’s the shocker: In this simple task, Glossophaga required more than 5000 trials before reaching a criterion of 85% correct responses [9], because they could not ignore which side it was presented on. And when they were tested in a new experimental room, they forgot what they learned about shape in the old room [9].
This is astounding because we know that flower bats use shapes a lot to identify flowers and find flower openings. In fact, the shapes of some bat-pollinated flowers evolved to signal bats.
Learned spatial cues can even be more powerful than cues that are instinctually attractive. Many neotropical flowers that are pollinated by bats produce a compound called dimethyl disulfide, which is powerfully and innately attractive to these bats [10]. If you take a test tube of dimethyl disulfide into a cage of captive Glossophaga that have never smelled it before in their life, they will fly over and stick their head into the test tube. They love it even without any previous experience.
But once Glossophaga learns a rewarded location, their spatial memory will still overshadow their use of dimethyl disulfide. I did this experiment myself [11]. I was surprised. If you move a feeder over a few feet, they will fly and hover at the blank spot on the wall where the feeder used to hang, before going over to the feeder clearly marked with rewarded smells and shapes.
The niche-specific cognitive strategy suggests that animal-eating bats should learn in a completely different way because prey don’t stay still. So sensory cues should be salient and learning food rewards based on sensory cues should be very easy for predatory bats. They should learn food-rewarded shapes or sounds faster than rewarded locations. And this seems to be true. Siemers [12] reported supportive evidence from a single insectivorous bat Myotis nattereri that quickly and easily learned to ignore location and associate shapes with food. They are the opposite of Glossophaga.
I copied and pasted the relevant part below because this paper is hard to get online. I just love how Bjorn Siemers turned a possible mere anecdote into a nice little study using careful observation and on-the-spot experimentation. People who say you can’t really learn anything from a sample size of one, take note. He reports [12]:
…a female M. nattereri lost its balance and slid into a white round bowl next to the cage. The bowl contained about 250 mealworms. The bat grabbed a mealworm and flew off. The bat returned in a few seconds and approached the bowl, striking its uropatagium [tail membrane] against the rim of the bowl. The bat repeated the approach flight three times, retrieving one or two more mealworms. I then moved the bowl in the flight tent and started videotaping the bat’s behavior. When the bat flew by the bowl at the new position, it hovered around it for 240 s without touching it. Over the next 60 min, the bat spent 650 s in eight 30 to 150 s long hover-and-attack- bouts above and around the bowl. The bat attacked the bowl 101 times by touching the inner or outer side of the rim with its uropatagium. When the bat touched the inner side of the bowl, it sometimes successfully caught a mealworm in the uropatagium…
 …I then removed the bowl and put three alive and moving mealworms onto the smooth cardboard surface where the bowl had been. The bat inspected the site for 100 s without landing or touching the surface or catching any mealworms. When I placed the round bowl (now without mealworms) back onto the cardboard, the bat attacked the empty bowl (13 times in 120 s). I filled the bowl again with mealworms and the bat attacked the bowl another 28 times (three hover-and-attack- bouts with total duration of 180 s). Subsequently I initiated a choice experiment whereby at the opposite edges of the cardboard surface, the round bowl (empty) was presented simultaneously with the square one, new to the bat but filled with about 250 live mealworms. The rustling mealworms were clearly audible at least to my human ear from where the bat hovered over the experimental set up. The bat was tested during two hover-and-attack-bouts in this choice experiment (duration of bouts 110 s and 130 s, respectively). The position of the bowls was exchanged between the two bouts. While 20 attacks were directed at the round, empty bowl, only two (unsuccessful) attacks occurred at the quadratic one with mealworms (Fig. 1). The bat significantly preferred the empty [round] bowl as a target over the square one (χ2 = 14.7, d.f. = 1, P < 0.001). In the next night, I again presented the round, empty bowl on the cardboard. The bat attacked three times in one 20 s hover-and-attack-bout and then lost interest
…I then removed the bowl and put three alive and moving mealworms onto the smooth cardboard surface where the bowl had been. The bat inspected the site for 100 s without landing or touching the surface or catching any mealworms. When I placed the round bowl (now without mealworms) back onto the cardboard, the bat attacked the empty bowl (13 times in 120 s). I filled the bowl again with mealworms and the bat attacked the bowl another 28 times (three hover-and-attack- bouts with total duration of 180 s). Subsequently I initiated a choice experiment whereby at the opposite edges of the cardboard surface, the round bowl (empty) was presented simultaneously with the square one, new to the bat but filled with about 250 live mealworms. The rustling mealworms were clearly audible at least to my human ear from where the bat hovered over the experimental set up. The bat was tested during two hover-and-attack-bouts in this choice experiment (duration of bouts 110 s and 130 s, respectively). The position of the bowls was exchanged between the two bouts. While 20 attacks were directed at the round, empty bowl, only two (unsuccessful) attacks occurred at the quadratic one with mealworms (Fig. 1). The bat significantly preferred the empty [round] bowl as a target over the square one (χ2 = 14.7, d.f. = 1, P < 0.001). In the next night, I again presented the round, empty bowl on the cardboard. The bat attacked three times in one 20 s hover-and-attack-bout and then lost interest
…After one accidental experience, an experimental bat learned to associate the round bowl with prey. The bat never attacked mealworms on the cardboard where the round bowl had been, and rarely did it look for mealworms in the square bowl that was unfamiliar to it as a feeding site. But the bat readily and repeatedly tried to retrieve prey from the round bowl, making attempts to take prey even when the bowl was empty. The evidence suggests that the bat was not perceiving the meal worms themselves but was operantly conditioned to the bowl as an indication of prey.
This finding was replicated and extended by Hulgard and Ratcliffe [13] who trained four additional bats of this species using rewarded styrofoam shapes hung from the ceiling. All the bats learned to associate a specific shape with nearby food rewards and their shape training even seemed to overshadow their use of spatial memory.
So we have a flower-visiting bat that takes just one trial to learn a location and 5000+ trials to learn a rewarded shape, because spatial cues overshadow shape cues. By contrast, we have an insect-eating bat that takes just one trial to learn a shape, and once learned, this interferes with later spatial learning.
In the lab I’m currently in, Rachel Page has done a decade of research on how frog-eating bats use different cues and combinations of cues under varying contexts for finding their prey. But what we still need is one large comparative study using the same design across more species, as has been done with this massive comparison of cognitive self-control in primates [14].
Even better than just testing use of different cues (spatial, visual, echo-acoustic shape, smell, etc) would be testing the association rates between different cues and their natural outcomes. For example, a flower bat should readily match a location with either a food reward or an “escape-from-danger” reward (i.e. learn a hiding spot). In contrast, an insect-eating bat might more quickly learn to associate a location as a hiding spot than as a place to get food. Experiments like this would test that associative learning is influenced not only by the cue salience of the learner (determined by the ecological challenges it faced during its evolutionary history) but also on the ecological validity of the association itself (certain associations, like taste-sickness, simply make more ecological sense).
References
1. Garcia, John, and Robert A. Koelling. “Relation of cue to consequence in avoidance learning.” Psychonomic Science 4.1 (1966): 123-124.
2. Macphail, Euan M., and Johan J. Bolhuis. “The evolution of intelligence: adaptive specializations versus general process.” Biological Reviews of the Cambridge Philosophical Society 76.03 (2001): 341-364.
3. Dunlap, Aimee S., and David W. Stephens. “Experimental evolution of prepared learning.” Proceedings of the National Academy of Sciences 111.32 (2014): 11750-11755.
4. Clayton, Nicola S., and John R. Krebs. “Memory for spatial and object-specific cues in food-storing and non-storing birds.” Journal of Comparative Physiology A 174.3 (1994): 371-379.
5. Brodbeck, David R. “Memory for spatial and local cues: A comparison of a storing and a nonstoring species.” Animal Learning & Behavior 22.2 (1994): 119-133.
6. Clare, Elizabeth L., et al. “Trophic niche flexibility in Glossophaga soricina: how a nectar seeker sneaks an insect snack.” Functional ecology 28.3 (2014): 632-641.
7. Winter Y, von Helversen O (2001) Bats as pollinators: foraging energetics and floral adaptations. In: Chittka L, Thomson J, editors. Cognitive ecology of pollination. Oxford: Oxford University Press.. 360 p.
8. Thiele J, Winter Y (2005) Hierarchical strategy for relocating food targets in flower bats: spatial memory versus cue-directed search. Anim Behav 69: 315–327.
9. Stich KP, Winter Y (2006) Lack of generalization of object discrimination between spatial contexts by a bat. J Exp Biol 209: 4802–4808.
10. Von Helversen, Otto, L. Winkler, and H. J. Bestmann. “Sulphur-containing “perfumes” attract flower-visiting bats.” Journal of Comparative Physiology A 186.2 (2000): 143-153.
11. Carter, Gerald G., John M. Ratcliffe, and Bennett G. Galef. “Flower bats (Glossophaga soricina) and fruit bats (Carollia perspicillata) rely on spatial cues over shapes and scents when relocating food.” PloS one 5.5 (2010): e10808.
12. Siemers, Björn M. “Finding prey by associative learning in gleaning bats: experiments with a Natterer’s bat Myotis nattereri.” Acta Chiropterologica 3.2 (2001): 211-215.
13. Hulgard, Katrine, and John M. Ratcliffe. “Niche-specific cognitive strategies: object memory interferes with spatial memory in the predatory bat Myotis nattereri.” Journal of Experimental Biology 217.18 (2014): 3293-3300.
14. MacLean, Evan L., et al. “The evolution of self-control.” Proceedings of the National Academy of Sciences 111.20 (2014): E2140-E2148.

Wonderful post Gerry.
LikeLike