This week, Julia Vrtilek passed her PhD Defense in the department of Evolution, Ecology and Organismal Biology at The Ohio State University. Members of my former lab at Ohio State were there and we had a nice but too brief "reunion". Her dissertation is entitled "Vocal convergence, responsiveness, and labeling in vampire bats". OSU Carter … Continue reading Congratulations Dr. Vrtilek!
Hiring a Research Coordinator
I am looking to hire: Research Coordinator (based in Panama) to (1) manage research and animal care for a vampire bat colony located at the Smithsonian Tropical Research Institute in Gamboa, Panama and (2) help with fieldwork in Panama and Costa Rica. I strongly encourage Panamanian Citizens to apply. Job description here.
Online talk and Psychology Seminar
I recently gave an online talk for a general audience entitled The Social Lives of Vampire Bats, hosted by Bat Conservation International. The video is below. https://www.youtube.com/embed/1e9_ZjUMfuo?si=6BqKKCV8eOZ-DSlC On Halloween Oct 31 at 12 noon, I'm also giving a talk entitled "Do Vampire Bats Have Friends?" for the Princeton Department of Psychology. LINK
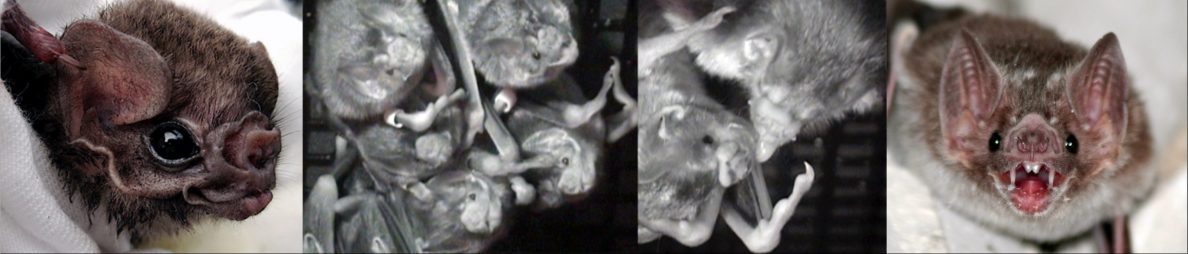
Lab updates: Fall 2025
The lab is growing! We have two new PhD students, Bryson and Nakul. Bryson Loflin received his Bachelors in Biology from UNC Chapel Hill, where he was mentored by Karin Pfennig and did research on exploratory behaviors in spadefoot toads. He also worked with Matthew Zipple in Mike Sheehan's Lab on social behavior of re-wilded … Continue reading Lab updates: Fall 2025
Join me on July 2 in “Exploring the social lives of bats”
I am giving an online talk, Exploring the social lives of bats, on Wednesday July 2 at 9am - 10 am EST. It is for a seminar series called "My Field for Dummies" hosted by GBatNet (Global Union of Bat Diversity Networks). To attend, please RSVP here: https://www.gbatnet.org/events/my-field-for-dummies-social-lives-of-bats/ Photo by Charles J. Sharp
Latest paper: A cryptic role for reciprocal helping in a cooperatively breeding bird
Latest paper, A cryptic role for reciprocal helping in a cooperatively breeding bird was published yesterday in the journal Nature. It's an analysis of 20 years of field observations of African superb starlings. The key discovery is that both related and unrelated birds were taking turns for each other as ‘breeders’ and ‘helpers’ across their … Continue reading Latest paper: A cryptic role for reciprocal helping in a cooperatively breeding bird
Latest paper: Long-term cooperative relationships among vampire bats are not strongly predicted by their initial interactions
Our latest paper reports on two experiments, the first conducted by former undergraduate Vi Girbino and the second by former postdoc Simon Ripperger. Both experiments asked the same question: Can we predict social bond formation in vampire bats from the way two bats interact at their first encounter? The reason we care about this question … Continue reading Latest paper: Long-term cooperative relationships among vampire bats are not strongly predicted by their initial interactions
The 2024 Ohio Bat Fest will be in Columbus, Ohio on October 19
The Carter Lab will hosting the 2024 Ohio Bat Festival in Columbus, Ohio on October 19. Sadly, this will be the last time the Ohio Bat Fest will be hosted by the Carter Lab. In 2025, we hope the Ohio Bat Fest will move to Cincinnati. Click here to learn more. We thank PhD Candidate Julia Vrtilek … Continue reading The 2024 Ohio Bat Fest will be in Columbus, Ohio on October 19
Seeking potential Princeton Postdoctoral Fellows
Please help me get the word out. I am seeking postdoctoral candidates to nominate for the Presidential Postdoctoral Research Fellows Program at Princeton University. The candidate could work on one of several ongoing projects involving cooperation and social bonding, depending on the candidate's background, but I also encourage more independent project ideas. Candidates will earn an … Continue reading Seeking potential Princeton Postdoctoral Fellows
Gerry starts HHMI Freeman Hrabowski Scholarship as an Associate Professor at Princeton University
As of September 1, I started my new position as an Associate Professor in the Department of Ecology and Evolutionary Biology at Princeton University. After a year-long delay, I am also finally starting my Freeman Hrabowski Scholarship with the Howard Hughes Medical Institute (HHMI). Both of these new positions come with a huge amount of support for … Continue reading Gerry starts HHMI Freeman Hrabowski Scholarship as an Associate Professor at Princeton University