Our latest paper here. Also some early press here. There are actually three recent papers on social networks and the benefits of network size in primates (by Laurent Brent and co-authors), songbirds (by Josh Firth and co-authors), and bats (by me and co-authors):
Family network size and survival across the lifespan of female macaques
by L. J. N. Brent, A. Ruiz-Lambides, M. L. Platt
Abstract: Two decades of research suggest social relationships have a common evolutionary basis in humans and other gregarious mammals. Critical to the support of this idea is growing evidence that mortality is influenced by social integration, but when these effects emerge and how long they last is mostly unknown. Here, we report in adult female macaques that the impact of number of close adult female relatives, a proxy for social integration, on survival is not experienced uniformly across the life course; prime-aged females with a greater number of relatives had better survival outcomes compared with prime-aged females with fewer relatives, whereas no such effect was found in older females. Group size and dominance rank did not influence this result. Older females were less frequent targets of aggression, suggesting enhanced experience navigating the social landscape may obviate the need for social relationships in old age. Only one study of humans has found age-based dependency in the association between social integration and survival. Using the largest dataset for any non-human animal to date, our study extends support for the idea that sociality promotes survival and suggests strategies employed across the life course change along with experience of the social world.
From the world’s longest-running field study of birds at Oxford…
Wild birds respond to flockmate loss by increasing their social network associations to others
by Josh A. Firth, Bernhard Voelkl, Ross A. Crates, Lucy M. Aplin, Dora Biro, Darren P. Croft, Ben C. Sheldon
Abstract: Understanding the consequences of losing individuals from wild populations is a current and pressing issue, yet how such loss influences the social behaviour of the remaining animals is largely unexplored. Through combining the automated tracking of winter flocks of over 500 wild great tits (Parus major) with removal experiments, we assessed how individuals’ social network positions responded to the loss of their social associates. We found that the extent of flockmate loss that individuals experienced correlated positively with subsequent increases in the number of their social associations, the average strength of their bonds and their overall connectedness within the social network (defined as summed edge weights). Increased social connectivity was not driven by general disturbance or changes in foraging behaviour, but by modifications to fine-scale social network connections in response to losing their associates. Therefore, the reduction in social connectedness expected by individual loss may be mitigated by increases in social associations between remaining individuals. Given that these findings demonstrate rapid adjustment of social network associations in response to the loss of previous social ties, future research should examine the generality of the compensatory adjustment of social relations in ways that maintain the structure of social organization.
Given their relevance, if these papers had come out earlier, I would have cited them!
Social bet-hedging in vampire bats
by Gerald G. Carter, Damien R. Farine, Gerald S. Wilkinson
Abstract: Helping kin or nonkin can provide direct fitness benefits, but helping kin also benefits indirect fitness. Why then should organisms invest in cooperative partnerships with nonkin, if kin relationships are available and more beneficial? One explanation is that a kin-limited support network is too small and risky. Even if additional weaker partnerships reduce immediate net cooperative returns, individuals extending cooperation to nonkin can maintain a larger social network which reduces the potential costs associated with losing a primary cooperation partner. Just as financial or evolutionary bet-hedging strategies can reduce risk, investing in quantity of social relationships at the expense of relationship quality (‘social bet-hedging’) can reduce the risks posed by unpredictable social environments. Here, we provide evidence for social bet-hedging in food-sharing vampire bats. When we experimentally removed a key food-sharing partner, females that previously fed a greater number of unrelated females suffered a smaller reduction in food received. Females that invested in more nonkin bonds did not do better under normal conditions, but they coped better with partner loss. Hence, loss of a key partner revealed the importance of weaker nonkin bonds. Social bet-hedging can have important implications for social network structure by influencing how individuals form relationships.
(here’s all the text copied below)
1. Introduction
When cooperative relationships require an investment of time or energy, individuals should invest preferentially in the partner yielding the greatest cooperative returns [1–3]. However, if cooperative relationships take time to develop and partners are not always available, then a strategy that focuses investments in the single most-profitable partnership is risky. When partner availability is unpredictable, a better strategy would diversify cooperative investments across more partners to reduce the potential costs of losing a key partnership. We call this strategy social bet-hedging. Like other forms of bet-hedging [4], this strategy can be advantageous even if it reduces average short-term returns.
Bet-hedging strategies avoid risk. Social bet-hedging is analogous to evolutionary bet-hedging, where phenotypes with less temporally variable reproductive success outbreed phenotypes yielding reproductive success that is higher on average but more temporally variable [4]. This occurs because optimizing growth rates (or returns on investment) requires increasing the geometric, rather than arithmetic, mean. An evolutionary bet-hedging strategy can maximize geometric mean fitness, even at the expense of a lower arithmetic mean fitness, by coping better with rare stressful conditions [4].
By spreading cooperative investments to more partners, social bet-hedging strategies can reduce the temporal variance in cooperative returns caused by changes in partner availability. Investing in new relationships can be beneficial even if this requires diverting time and energy away from the most-profitable cooperative relationship which yields the greatest inclusive fitness return rate (e.g. the strongest reciprocator or closest kin).
Social bet-hedging might explain why female common vampire bats (Desmodus rotundus) that have strong reciprocal food-sharing relationships with close kin still regurgitate food to other nonkin [5–7]. Vampire bats are susceptible to starvation and depend on a network of food-sharing partners to feed them after unsuccessful foraging nights. The strongest, most reliable, and most balanced food-sharing bonds develop between mothers and daughters, but even for these close kin, the direct fitness benefits of food sharing might exceed the indirect fitness benefits [5–10]. The best known predictor of sharing rates within familiar pairs is not kinship, but the reciprocal rate of sharing [5,8]. If feeding close kin yields reciprocal sharing benefits that are equal or greater to feeding nonkin, why invest in nonkin bonds?
Sharing only with kin could be risky because relatives can be lost for various reasons. A starved female with only one or a few close maternal kin in her food-sharing network might not find her primary close kin donor, for example, if this partner also failed to feed or switched to a different roost on that night—which happens frequently [10]. To compensate for this risk, a social bet-hedging female would foster new bonds by diverting some of her social time and energy away from mothers and daughters and towards other females. Even if each of these additional partners is less related and reciprocates less, this strategy could dramatically increase long-term survival by reducing the risk of failing to find a primary donor when in dire need.
To test this idea, we quantify the impact, in terms of total food received, of removing a past key food donor for individual bats in need. Previously, Carter & Wilkinson [6] demonstrated that females that fed more nonkin females in previous years subsequently received more food in the absence of this key donor (see Methods), but this finding could simply mean that better-connected bats always receive more food. Here, we extend our analysis of this experiment to show that, as predicted by social bet-hedging, helping more nonkin did not increase food received when key donors were available, but it reduced the negative impact on food received when a key donor was removed.
2. Methods
We used data from a previous experiment [6], where a female subject was isolated and fasted for 24 h, then reintroduced to a captive colony of 27–34 individually marked conspecifics to measure food donated by each groupmate. Mean dyadic donation rates were calculated from 1337 dyadic regurgitation observations among 14 captive females using 91 fasting trials over a 4-year period (see electronic supplementary material, [6]). Relatedness was estimated using maternal pedigree and 19 microsatellite markers (see [6]). For each female, a unique key donor with a strong history of food sharing was selected for temporary removal; key donors were either the subject’s highest-ranking donor (nine cases), second-highest ranking donor (four cases), or a lower-ranking donor but the highest-ranking recipient (one case) (see electronic supplementary material, [6]). During two control trials, a female that had never fed the subject was excluded by either removing it or fasting it on the same night. During three subsequent test trials, the subject’s key donor was similarly excluded [6]. A previous analysis showed that bats that fed more nonkin females in past years received more food during test trials [6], but the social bet-hedging hypothesis predicts that this relationship should be most important when key donors are removed, not when they are present.
Here, we fitted linear models to predict the amount of food received with and without key donors present as well as the change in total food received (difference in food received per trial) when key donors were removed. We included the number of nonkin females fed in the past 4 years to represent investment in the size of a social support network. We did not include the number of kin fed because this depended on the number of kin available. We also did not include the number of males fed because stable bonds in the wild are female–female. To control for sampling bias, we included the control variable opportunity to donate, which is the number of trials where the subject could have fed another bat (see electronic supplementary material). The distribution of residuals did not deviate from normal (Shapiro Wilk’s test: W = 0.98, p = 0.95). To visualize results, we plotted mean food received against residual past sharing to nonkin females—the residuals from a regression of the number of unrelated females fed on number of opportunities to donate (to control for the latter).
3. Results
Under typical conditions, when key donors were present, the number of female nonkin fed in previous years did not predict the amount of food received; instead the trend was negative (figure 1a;R2= 0.43, β = −52.4, t = −1.81, p = 0.155). However, feeding more female nonkin did predict receiving more food later when a female’s key donor was absent (figure 1b;R2 = 0.56, β = 56.0, t = 3.68, p = 0.004). A bat’s proclivity to invest in female nonkin therefore predicted the change in total food she received when key donors were removed (R2 = 0.58, F2,11 = 7.54; β = 108.4, t = 3.48, p = 0.005; figure 1c). Females that fed more female nonkin coped better with partner removal. This result was robust to several variations in the analysis (see electronic supplementary material).
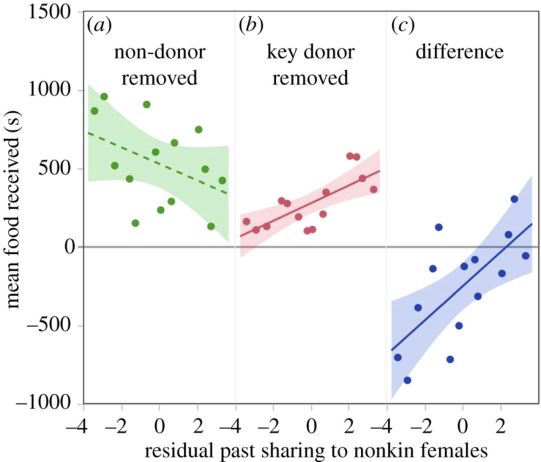
Figure 1.
Bats with a higher propensity to help unrelated females suffered smaller losses in total food received when a key donor was removed as a potential donor. Proclivity to feed more nonkin females (x-axis) did not positively correlate with food received when a non-donor was absent (a) but it did when the key donor was absent (b). Feeding more unrelated females predicted smaller reductions in food received when the key donor was removed (c). Shading shows 95% CI of the slope.
4. Discussion
Our results support the social bet-hedging hypothesis. By helping nonkin, individuals appear to maintain a wider support network than would be possible through only helping close kin. This suggests that female vampire bats can reduce the costs of losing a key donor by ‘not putting all their eggs in one basket’.
The social bet-hedging hypothesis makes three key assumptions. First, it assumes that individuals shift cooperative investments to and from individuals based on their relative cooperative returns, as predicted by reciprocity and biological market theory (e.g. models of partner control and partner choice) [1–3].
Second, it assumes not only that there are fitness benefits to having both more cooperative partners and stronger relationships [11–17], but also that individuals often face a trade-off between investing in relationship quantity versus quality (strength). If cooperative relationships require continuous investment, then merely increasing the number of weak connections can reduce overall cooperative returns, just as increasing offspring production at the expense of offspring quality does not reliably increase fitness [18]. On the other hand, strengthening each relationship can come at the expense of relationship quantity, so individuals might therefore divert investments towards partners that yield lower indirect fitness or reciprocal returns simply to create more relationships.
Third, social bet-hedging only makes sense if lost cooperative partnerships cannot be replaced instantly and effortlessly (as evidenced by figure 1a). Backup partners must already be in place. Social bet-hedging therefore assumes that new relationships require time and energy to develop. This seems true for food-sharing vampire bats [5–10].
Social bet-hedging may also exist for other cooperative behaviours. For example, female baboons increase their social grooming rates and groom more partners after the death of a close female relative [19], suggesting that investments in more relationships can help to compensate for the loss of a key social partner. In humans, although relationship quality is better than relationship quantity at predicting received social support [20], people appear to benefit from a greater number of weaker friendships in environments where friends are more likely to leave [21].
Many models of cooperation focus on pairwise interactions (e.g. [2]), but cooperative ‘exchange rates’ are determined by the supply and demand of cooperative services and partners—properties of the larger social network [22]. Many cooperative species might allocate cooperative investments across several partners and compare the varying return rates from each [3,22]. It remains unclear, however, if or how different social animals balance the quality and quantity of social ties. By influencing the number and strength of connections in a social network, strategies like social bet-hedging can both shape, and be shaped by, social network structure.
Ethics: All procedures were approved by the University of Maryland Institutional Animal Care and Use Committee (Protocol R-10-63).
Data accessibility: The data supporting this article have been uploaded as part of the electronic supplementary material.
Authors’ contributions: G.G.C. and D.R.F. conceived the analysis, G.G.C. carried out the analysis, and D.R.F. and G.S.W. advised the analysis. G.G.C. drafted the manuscript; D.R.F. and G.S.W. revised it critically for important intellectual content. All authors gave final approval of the version to be published, and agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Funding: Work by G.G.C. was supported by a Ford Predoctoral Fellowship, a Dissertation Improvement Grant from the National Science Foundation (IOS-1311336), and grants from the American Society of Mammalogists and Animal Behavior Society.
Acknowledgements: We thank the Organization for Bat Conservation for their extraordinary support. Ronald Noë, Rachel Crisp, Julia Vrtilek and two anonymous reviewers provided comments that improved the manuscript.
References
- [1] Trivers, R.L. 1971 The evolution of reciprocal altruism. Quarterly Review of Biology. 46, 35-57.
- [2] Axelrod, R. & Hamilton, W.D. 1981 The evolution of cooperation. Science 211, 1390-1396.
- [3] Noë, R. & Hammerstein, P. 1994 Biological markets: supply and demand determine the effect of partner choice in cooperation, mutualism and mating. Behavioral Ecology and Sociobiology 35, 1-11.
- [4] Philippi, T. & Seger, J. 1989 Hedging one’s evolutionary bets, revisited. Trends in Ecology & Evolution 4, 41-44.
- [5] Carter, G.G. & Wilkinson, G.S. 2013 Food sharing in vampire bats: reciprocal help predicts donations more than relatedness or harassment. Proceedings of the Royal Society of London B 280, 20122573.
- [6] Carter, G.G. & Wilkinson, G.S. 2015 Social benefits of non-kin food sharing by female vampire bats. Proceedings of the Royal Society of London B 282, 20152524-20152524.
- [7] Wilkinson, G.S. 1984 Reciprocal food sharing in the vampire bat. Nature 308, 181-184.
- [8] Carter, G.G. & Wilkinson, G. 2013 Does food sharing in vampire bats demonstrate reciprocity? Communicative and Integrative Biology 6, e25783. (doi:10.4161/cib.25783).
- [9] Wilkinson, G.S. 1988 Reciprocal altruism in bats and other mammals. Ethology and Sociobiology 9, 85-100.
- [10] Wilkinson, G.S. 1985 The social organization of the common vampire bat: I. Pattern and cause of association. Behavioral Ecology and Sociobiology 17, 111-121.
- [11] Kokko, H., Johnstone, R.A. & Clutton-Brock, T.H. 2001 The evolution of cooperative breeding through group augmentation. Proceedings of the Royal Society of London B 268, 187-196.
- [12] Seyfarth, R.M. & Cheney, D.L. 1984 Grooming, alliances and reciprocal altruism in vervet monkeys. Nature 308, 541-543.
- [13] Seyfarth, R.M. & Cheney, D.L. 2012 The evolutionary origins of friendship. Annual Review of Psychology 63, 153-177.
- [14] Seyfarth, R.M., Silk, J.B. & Cheney, D.L. 2014 Social bonds in female baboons: the interaction between personality, kinship and rank. Animal Behaviour 87, 23-29.
- [15] Silk, J.B., Beehner, J.C., Bergman, T.J., Crockford, C., Engh, A.L., Moscovice, L.R., Wittig, R.M., Seyfarth, R.M. & Cheney, D.L. 2009 The benefits of social capital: close social bonds among female baboons enhance offspring survival. Proceedings of the Royal Society of London B 276, 3099-3104.
- [16] Silk, J.B., Beehner, J.C., Bergman, T.J., Crockford, C., Engh, A.L., Moscovice, L.R., Wittig, R.M., Seyfarth, R.M. & Cheney, D.L. 2010 Strong and consistent social bonds enhance the longevity of female baboons. Current Biology 20, 1359-1361.
- [17] Wittig, R.M., Crockford, C., Lehmann, J., Whitten, P.L., Seyfarth, R.M. & Cheney, D.L. 2008 Focused grooming networks and stress alleviation in wild female baboons. Hormones and Behavior 54, 170-177.
- [18] Smith, C.C. & Fretwell, S.D. 1974 The optimal balance between size and number of offspring. The American Naturalist 108, 499-506.
- [19] Fruteau, C., Voelkl, B., van Damme, E. & Noë, R. 2009 Supply and demand determine the market value of food providers in wild vervet monkeys. Proceedings of the National Academy of Sciences USA 106, 12007-12012.
- [20] Engh, A.L., Beehner, J.C., Bergman, T.J., Whitten, P.L., Hoffmeier, R.R., Seyfarth, R.M. & Cheney, D.L. 2006 Behavioural and hormonal responses to predation in female chacma baboons (Papio hamadryas ursinus). Proceedings of the Royal Society of London B 273, 707-712.
- [21] Franks, H.M., Cronan, T.A. & Oliver, K. 2004 Social support in women with fibromyalgia: Is quality more important than quantity? Journal of Community Psychology 32, 425-438.
- [22] Oishi, S. & Kesebir, S. 2012 Optimal social-networking strategy is a function of socioeconomic conditions. Psychological Science, 0956797612446708.
Text Supplement
Supplement to methods
For each female test subject, we selected a unique key donor. All targeted key donor pairs were symmetrical such that if bat A was the key donor for bat B, then B was the targeted key donor for A. This was possible because if bat A was one of the most frequent donors for B, then B was typically one of the most frequent donors for A. The key donor was the subject’s highest-ranking donor, i.e. the bat that gave it the most food, in nine cases (including three mothers, four daughters, another relative, and one nonrelative). The key donor was the subject’s the second-highest-ranking donor in four cases (including one mother, another relative, and two nonrelatives). However, in one case (one nonrelative), the key donor was the highest-ranking recipient for the subject, but only the ninth-highest-ranking donor. In each trial, a possible donor was excluded by either removing it or fasting it on the same night, and this occurred on five occasions separated by at least 7 days.
The control variable opportunity to donate explained 79% of the variation in the observed number of partners fed (F(1,13)=48, p<0.0001), and it also predicted the number of nonkin females fed (R2=0.31, F(1,13)=5.5, p=0.038). Opportunity to donate was determined by a bat’s presence or absence in the home cage during past fasting trials, which was influenced by age (Spearman’s correlation with age: 0.58; p=0.03) and the bat being haphazardly excluded from some past fasting trials based on sickness, injury, or recent birth of a pup.
Supplement to results
When controlling for opportunity to donate, the effect of the number of nonkin females fed on the change in food received remained the same after excluding three cases where key donors failed to donate (R2=0.64, n=11, F(2,8)=7.1, nonkin females fed: beta=115.5, t=3.16, p=0.0134). The effect was also detected when using nonkin females fed per opportunity to donate as the predictor (R2=0.29, n=14, F=4.79, p=0.0490). When we added key donor kinship as a factor (kin or nonkin) and the interaction between kinship and nonkin females fed as predictors in the model, we found no interaction effect (p=0.73). When this interaction term was removed, we found that nonkin females fed (beta=108.2, t=3.30, p=0.008), but not kinship (p=0.95), predicted the change in food received.
Supplement to discussion
Social bet-hedging can be seen as a strategy for balancing two potentially contradicting strategies: group augmentation, defined as investing in group members to increase group size, i.e. partner quantity (Kokko et al. 2001, Kingma et al. 2014), and reciprocity, defined as investing preferentially in specific partners that provide the best reciprocal returns, i.e. partner quality (Trivers 1971, Carter 2014). Imagine you are the ideal Darwinian primate, deciding how much to groom each member of your group to maximize cooperative returns and hence inclusive fitness. For simplicity in this case, assume all kinship is equal. Each groupmate will later provide you with food, if you have groomed them sufficiently, and each group member might differ in the amount of food they would feed you per unit of grooming received. Assume that return rates are tracked by some form of emotional scorekeeping, and that you therefore have full knowledge of which relationships provide the greatest return rates, assuming those individuals are present. There are countless ways to divide your time and energy. You could spend 100% of your grooming effort on your best food provider. You could allocate 85% to her and 10% to the next best and 5% to the third best. You could split your grooming effort equally among all group members. It’s easy to see the analogy with betting or financial investing. The optimal strategy will depend on the distribution of food-sharing probabilities and amounts (Kelly 1956). If one partner provides the highest return rate, then you should simply invest everything in that partner, i.e. a partner choice strategy. On the other hand, if all groupmates are equally likely to provide food and to provide equal amounts of food, then you should simply maximize the number of partners that are alive or available to you, i.e. group augmentation. However, if partner return rates or availability vary over time, then “social bet-hedging” becomes relevant.
The need for social bet-hedging also depends on the timescale of reciprocal relationship formation: how much grooming does it take to create a relationship that starts benefiting you with food sharing? If social grooming can immediately lead to food sharing in the next minute, then social bet-hedging is not necessary, because when food is needed, you can immediately divert social grooming to the current best partner. If, however, a food-sharing relationship requires several days of previous grooming investment, then you should already be grooming many different partners, in case they can become future food providers in a time of need. This form of social bet-hedging requires long-term contingency embedded within complex, stable, multi-benefit relationships, so it is most likely to be found in primate or primate-like societies composed of stable individualized cooperative relationships. It would be particularly interesting to test the extent to which subconscious decisions to invest in relationship quantity or quality influence human social networks of collaboration or friendship.
Supplement references
- Carter GG. 2014. The reciprocity controversy. Animal Behavior and Cognition 1:368-386.
- Kelly JL. 1956. A new interpretation of information rate. Bell System Technical Journal 35:917-926.
- Kingma SA, Santema P, Taborsky M, and Komdeur J. 2014. Group augmentation and the evolution of cooperation. Trends in Ecology and Evolution 29:476-484.
- Kokko H, Johnstone RA, and Clutton-Brock TH. 2001. The evolution of cooperative breeding through group augmentation. Proceedings of Royal Society London B 268:187-196.
- Trivers RL. 1971. The evolution of reciprocal altruism. Quarterly Review of Biology 46:35-57.

One thought on “Latest paper: social bet-hedging in vampire bats (and two other recent and related studies)”